
degradation of the implant. Multinuclear cells were followed by
osteoblasts, fibroblasts and new blood vessels. HA fragments were
surrounded by newly formed bone and multinuclear cells. Osteocytes
were enclosed in newly formed bone that was encircled by osteoblasts
and bone lining cells. High amount of blood vessels, fibrous tissue and
the formation of bone marrow was observed in the lacunae between
the HA fragments.
The HA/col-1 implants were also free of surrounding inflammatory
tissue reaction but revealed less fragmentation compared to
HA-implants (Figure 3). Most multinuclear cells were found on the
surface of the implant and only a few of themwere penetrating into the
implant. A small rim of new bone covered by osteoblasts was also
localised on the surface of implant and fragments. The implant was
surrounded by fibrous tissue. The HA/col-1 implants were more
fragmented in the iliac crest than in the vertebrae as described for the
HA implants.
Immuno-, enzymehistochemistry and in situ hybridization
Osteoblasts were identified using immunohistochemistry with an
antibody against osteocalcin and
in situ
hybridization for connexin-43
(Figure 4). Samples with HA implant showed a higher amount of
osteoblasts than samples with HA/col-1 implant. In the controls with
empty defects only a few osteoblasts were observed at the host bone
interface. In addition to osteoblasts, osteocytes surrounded by calcified
bone matrix showed also a positive connexin-43 labelling.
Newly formed bone was detected by collagen-type I immunohis-
tochemistry. High amounts of collagen-I immunoreactivity around
fragments of HA implants were found. TRAP could be seen within the
cytoplasm of mononuclear macrophages and multinucleated cells. The
same cell types were also positively stained for CD68 immunohisto-
chemistry. Osteoclast-like cells were localized along the surfaces of HA
particles which had not been covered by newly formed bone and
within the granulation tissue. eNOS immunoreactivity labelled
sprouting endothelial cells as well as osteoclast-like cells where the
staining was localized in the resorbing region. This labelling also
showed that newly formed blood vessels were localized in direct
vicinity of resorbing multinuclear cells. In controls with empty defects,
immunohistochemistry, TRAP and connexin-43 in-situ hybridization
no positive staining was detected.
Transmission electron microscopy
Transmission electron microscopy showed that multinuclear cells
localized at the interface of both the HA and HA/col-1 implants were
able to incorporate degraded implant material particles (Figure 5).
Multinuclear cells localised at the HA and HA/col-1 interface showed
osteoclast-like properties such as exhibition of several nuclei and usual
cell organelles and formation of sealing zones at the circumference of
the ruffled border. At the sealing zone, pseudopodia-like plasmapro-
trusions were formed to anchor the cell to the implant. Those
osteoclast-like cells formed several short but wide plasma protrusions
on the apical side. In the HA/col-1 group, osteoclast-like cells exhibited
frequently more cell nuclei but the sealing zone was less developed
compared to the HA group. Frequently, remnants of the HA implant
were found between ruffled borders invaginations in the cytoplasm
and in phagosomes but not in the nuclei.
Discussion
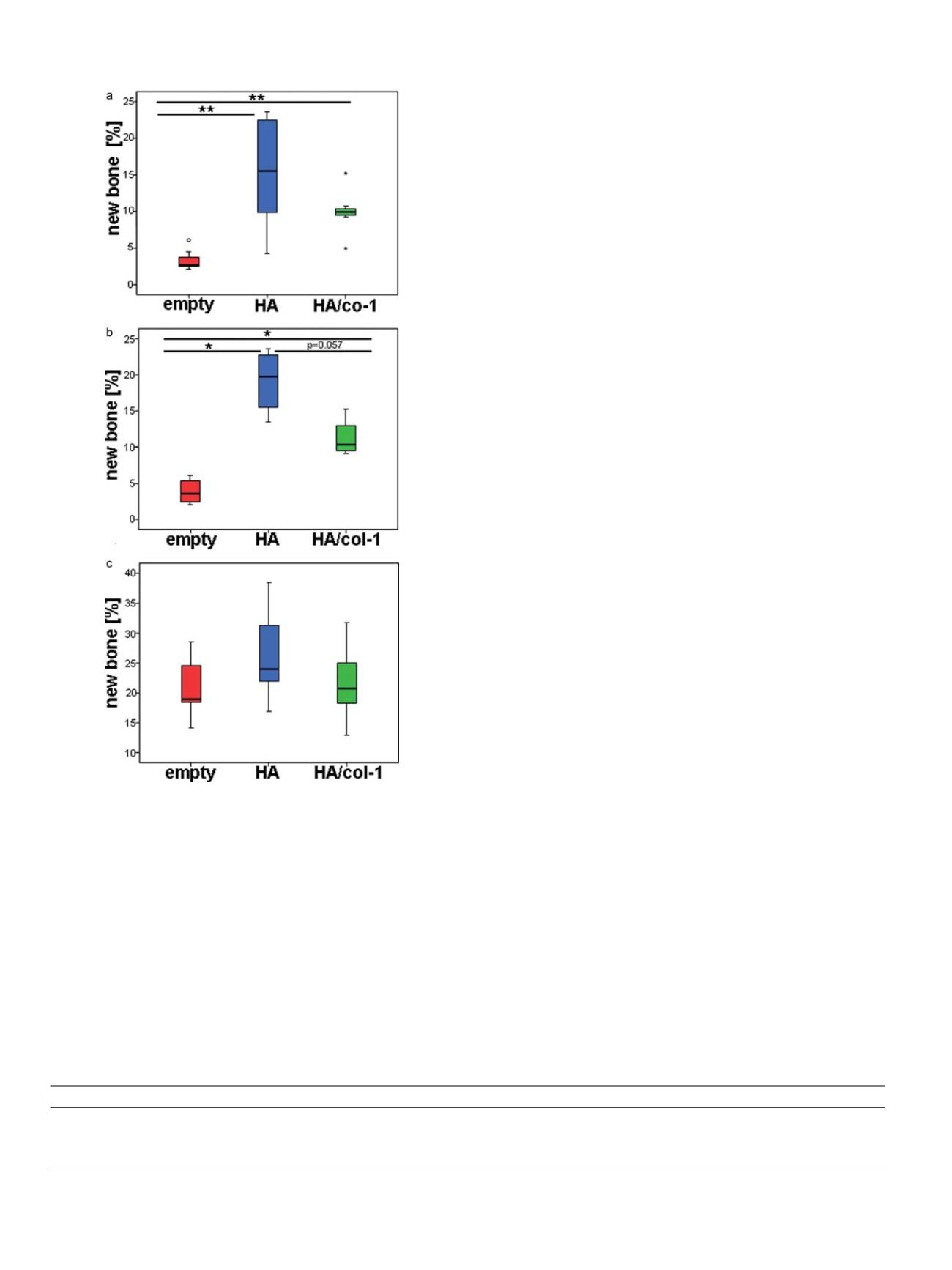
The current study revealed a statistically significant higher new
bone formation in the defect region of the HA and the HA/col-1 group
including all anatomical sites compared to the empty defect group in
this osteoporotic animal model in Chinese mountain goats by
Table 2
Histomorphometric parameters evaluated by HR-pQCT expressed in mean ± SD, shown with p-values of Kruskal-Wallis test between groups.
BMD (mmHA)
BV/TV (1)
Conn.D. (1/mm
3
)
Tb.N (1/mm)
Tb.Th(mm)
Tb.Sp(mm)
Empty
618.8 ± 29.0
0.32 ± 0.22
2.09 ± 1.41
1.86 ± 0.45
0.32 ± 0.09
0.59 ± 0.17
HA
650.3 ± 19.3
0.58 ± 0.10
3.42 ± 1.26
2.40 ± 0.14
0.44 ± 0.11
0.41 ± 0.04
HA/Col-1
639.6 ± 27.8
0.42 ± 0.11
4.62 ± 2.45
2.47 ± 0.27
0.36 ± 0.08
0.42 ± 0.06
p-value
0.067
0.008*
0.034*
0.002*
0.055
0.005*
*
p
< 0.05.
Fig. 2.
Histomorphometrical results for the defect region including all anatomical
sites (
a
), for lumbar vertebrae defects (
b
) and for the interface region with a 1 mm
distance to the initially created defect including all defects (
c
). (*p < 0.05, **p < 0.01)
V. Alt et al. / Injury, Int. J. Care Injured 47S2 (2016) S58
–
S65
S62


















