
histomorphometry. There were no significant differences between the
HA and the HA/col-1 group for this overall evaluation. HA could also
show enhanced new bone formation for site specific analysis for
vertebrae defects compared to the empty defect group. This confirms
findings of other
in vivo
studies on the osteoconductive properties
of the used nanocrystalline hydroxyapatite [2
–
10] which can be
explained by the almost identical calcium/calcium phosphate ratio of
human hydroxyapatite to the 1,67 ratio of this biomaterial [20]. Missing
differences between the HA- and the HA/col-1 group suggest that the
idea of enhancing adhesiveness of the biomaterial to osteoblasts
via transmembrane integrin receptors via collagen-type I failed in the
current study. However, it is difficult to assess if another HA/col-type-I
ratio or other modifications might have had an effect which remains
a question for potential further studies.
Histology revealed good biocompatibility with the absence of
inflammatory reactions both for HA- and HA/col-type I implants
confirming the results of other authors in physiological bone status
in maxillofacial [6] and long bone defects [7
–
8]. Analysis of the
degradation behaviour revealed higher fragmentation in the HA-
compared to the HA/col-type I group with multinuclear osteoclast-like
cells penetrating into the defect. In both groups, a considerable amount
of implant remnants could be observed as HA- or HA/col-type I
fragments that were surrounded by newly formed bone within the
defect after 6 weeks suggesting an incomplete degradation of the two
materials after 6 weeks. This is in line with findings from authors that
also reported incomplete resoprtion of this nanocrystalline HA after
several weeks [5,8,9]. Fragmentation was shown by transmission
electron microscopy to be caused by multinuclear osteoclast-like cells
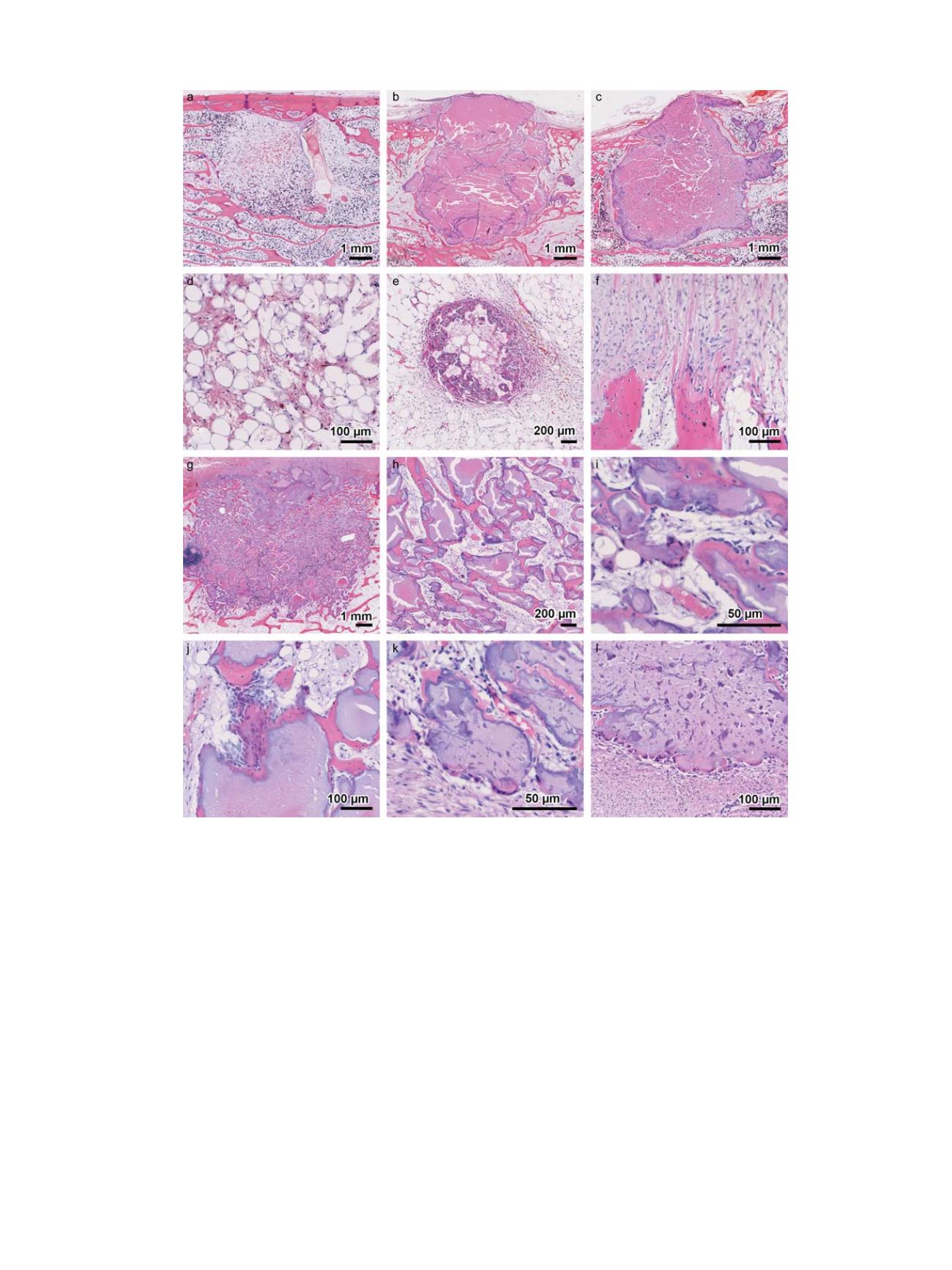
Fig. 3.
Histological analysis of defects in the vertebrae of the lumbar spine of the empty defect group (
a
), HA implant (
b
), and HA/col-1 implant (
c
) group. Empty defects were
mostly filled with bone marrow (
d
). Some empty defects showed an accumulation of multinucleated body giant cells and macrophages that were arranged as a circle with
centred fatty bone marrow (
e
). Host bone trabeculae at the edge of empty defects were covered with osteoblasts forming new collagen fibers into direction of the defect (
f
).
Overview of iliac crest defect filled with HA (
g
) with high fragmentation as shown in (
h
) in higher magnification. HA fragments were surrounded by newly formed bone
covered with osteoblasts and in addition by some multinucleated macrophages (
i
). Accumulation of active osteoblasts at the interface of a HA fragment (
j
). Higher magnification
of a HA/col-1 implant with high fragmentation but fragments were covered only with a small discontinuous rim of osteoid and osteoblasts (
k
), and a high number of multinu-
cleated foreign body giant cells (
l
).
V. Alt et al. / Injury, Int. J. Care Injured 47S2 (2016) S58
–
S65
S63


















